Synthesis and properties of geometrical 4-diarylmethylene analogs of the green fluorescent protein chromophore†
Abstract
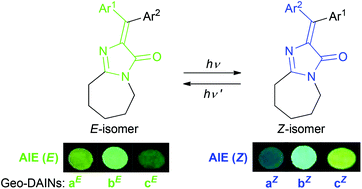
We have developed a novel analog of the GFP chromophore: geo-DAIN. Since geo-DAIN is equipped with an E/Z-photoisomerizable geometrical diarylmethylene moiety instead of benzylidene of the GFP chromophore, different-colored reversible emissions are expected. We synthesized geo-DAIN by a condensation with methyl imidate and N-(diarylmethylene)glycinate. We found the emission from geo-DAIN to be different from that of benzylidene-type analogs; in the powder state, the E- and Z-isomers of geo-DAIN emitted different fluorescence colors.


 Please wait while we load your content...
Please wait while we load your content...