Synthesis of a dimeric 3α-hydroxy-7α,12α-diamino-5β-cholan-24-oate conjugate and its derivatives, and the effect of lipophilicity on their anion transport efficacy†
Abstract
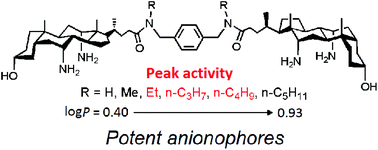
A dimeric 3α-hydroxy-7α,12α-diamino-5β-cholan-24-oate conjugate and its derivatives having alkyl chains of varying length from methyl to n-pentyl groups on the amido bonds were synthesized and fully characterized on the basis of NMR (1H and 13C) and ESI MS (LR and HR) data. Their transmembrane anion transport activities were investigated in detail by means of a chloride ion selective electrode technique and the pyranine assay. The data indicate that this set of compounds is capable of promoting the transmembrane transport of anions, presumably via an anion exchange process and a mobile carrier mechanism. Detailed kinetic analysis on the data obtained from both chloride efflux and pH discharge experiments reveals that an optimum log P range may exist for the transport effectiveness in terms of both k2/Kdiss and EC50 values. The present finding highlights the importance of high anionophoric activity in clarifying the effect of lipophilicity on ion-transport effectiveness.


 Please wait while we load your content...
Please wait while we load your content...