The chemistry of ynol and thioynol ethers
Abstract
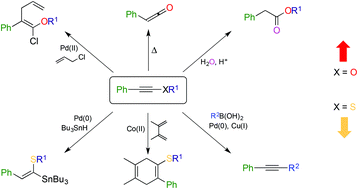
Alkynyl ethers and alkynyl thioethers (‘ynol ethers’ and ‘thioynol ethers’) are appealing building-blocks in synthetic chemistry due to their ease of manipulation and predictable reactivity. Until recently however, their potential has remained underexploited due to difficulties in preparation and isolation. Although recent advances in synthetic chemistry have highlighted various applications for ynol ethers, the equivalent thioynol examples have been rather less exploited despite a unique and fascinating reactivity profile. Although superficially the chemistry of alkynyl ethers and their sulfide counterparts are similar, close examination of their chemistry reveals important differences which can be exploited by the synthetic chemist. This review will examine the preparation of both classes of compound and examine their reactivity to highlight their powerful synthetic applications. Particular focus will be made of thiynol ethers whose chemistry exhibits some fascinating differences compared to their oxygen counterparts and have immense untapped potential for synthetic chemistry.

 Please wait while we load your content...
Please wait while we load your content...