An efficient strategy to enhance binding affinity and specificity of a known isozyme inhibitor†
Abstract
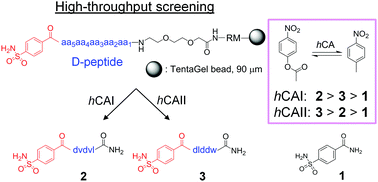
The binding profile of a known inhibitor, benzenesulfonamide, against a family of carbonic anhydrase isozymes was efficiently enhanced via high-throughput screening of customized combinatorial one-bead-one-compound peptide libraries modified with the inhibitor molecule. The screening of the conjugate libraries recognized subtle variations in the microenvironments of the target enzyme and thus facilitated the identification of short peptide sequences that bind selectively to a close proximity of the active site. The identified peptide portions contributed significantly to the overall binding of the conjugate peptides with greatly enhanced affinity as well as improved specificity towards the target isozyme. The interactions between the inhibitors and the isozymes were validated by surface plasmon resonance (SPR), pull-down assay and enzymatic activity measurement. This high-throughput approach proved useful and efficient to enhance the binding profile of known inhibitors and may apply to developing effective inhibitors for a wide range of isozyme families.

 Please wait while we load your content...
Please wait while we load your content...