New C16 fatty-acid-based oxylipin pathway in the marine diatom Thalassiosira rotula
Abstract
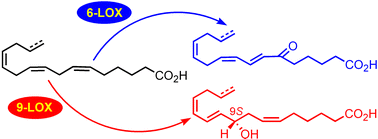
An unprecedented series of C16 oxylipins (1–8) has been characterized from the marine diatom Thalassiosira rotula. Absolute stereochemistry of the major alcohols 2 and 3 was determined to be 9S by spectroscopic and chemical methods. All the described products are formally derived by unprecedented enzymatic oxidation of C16 fatty acids. Conversion of hexadeca-6,9,12-trienoic acid (C16:3 ω-4) into 3 unequivocally established the occurrence of (at least) a specific 9S-oxygenase activity. To the best of our knowledge, the present data reveal for the first time the existence of an organic network of oxygenase-mediated transformations that require C16 fatty acids as substrates in living cells.

 Please wait while we load your content...
Please wait while we load your content...