Single Ni atoms with higher positive charges induced by hydroxyls for electrocatalytic CO2 reduction†
Abstract
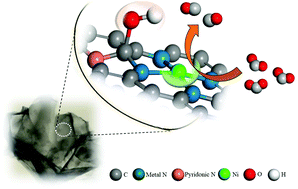
To promote the faradaic efficiency of the electrocatalytic CO2 reduction reaction (CO2RR) with low-cost catalysts, single Ni atoms with higher positive charges induced by hydroxyls were proposed to form an atomically dispersed Ni–N4 structure in a cheap honeycomb-like carbon matrix for electrocatalytic CO2 reduction. Extended X-ray absorption fine structure spectroscopy, aberration-corrected High-angle annular dark-field scanning transmission electron microscopy and X-ray photoelectron spectroscopy measurements confirmed that the active-center structure consists of single Ni atoms and the adjacent hydroxyl via hydrothermal treatment (H-Ni/NC). Density functional theory calculations indicated that the isolated Ni atoms with higher positive charges induced by the hydroxyl decreased the free energy of the rate-limiting step to 1.05 eV for the CO2RR. The faradaic efficiency (FE) of CO2 reduction into CO was ≥88.0% over the H-Ni/NC catalyst in the potential range of −0.5 to −0.9 V (vs. RHE). The peak CO FE reached 97% at −0.7 V due to the synergistic effect between the unsaturated Ni–N4 active sites and the hydroxyl species.


 Please wait while we load your content...
Please wait while we load your content...