in situ engineered ultrafine NiS2-ZnS heterostructures in micro–mesoporous carbon spheres accelerating polysulfide redox kinetics for high-performance lithium–sulfur batteries†
Abstract
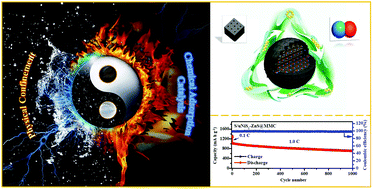
Host materials that can physically confine and chemically adsorb/catalyze lithium polysulfides (LiPSs) are currently receiving intensive research interest for developing lithium–sulfur (Li–S) batteries. Herein, a novel host material made of micro–mesoporous carbon nanospheres (MMC NSs) with well-dispersed ultrafine NiS2-ZnS (uNiS2-ZnS) heterostructures is synthesized for the first time via a simple in situ sulfuration process. The uNiS2-ZnS/MMC materials achieve the synergistic effect of physical confinement and the efficient chemical adsorption/catalysis of LiPSs through a micro–mesoporous structure and well-dispersed uNiS2-ZnS heterostructures. In addition, compared with bulk heterostructured materials, the uNiS2-ZnS heterostructures greatly enhance the adsorption and catalytic ability toward LiPSs because the catalysis interface effect and naturally formed in-plane interfaces can be magnified by the ultrafine dispersed nanoparticles. As a result, the prepared uNiS2-ZnS/MMC-S cathodes exhibit outstanding rate capacity (675.5 mA h g−1 at 5.0C) and cyclic stability (710.5 mA h g−1 at 1.0C after 1000 cycles with a low capacity decay of 0.033% per cycle). This work provides a certain reference for the application of heterostructured materials in Li–S batteries.


 Please wait while we load your content...
Please wait while we load your content...