Interfacial synergy between dispersed Ru sub-nanoclusters and porous NiFe layered double hydroxide on accelerated overall water splitting by intermediate modulation†
Abstract
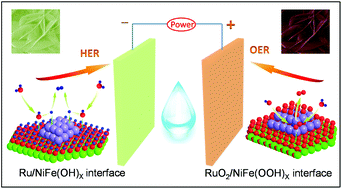
Construction of an efficient bifunctional electrocatalyst through a rational interface-engineering strategy to optimize the adsorption energy of H* and OH* species at the atomic/molecular level is of great importance for water splitting. Although conventional NiFe layered double hydroxide (LDH) shows excellent performance for alkaline oxygen evolution reactions (OERs), it shows extremely poor activity toward hydrogen evolution reactions (HERs) due to weak hydrogen adsorption and sluggish kinetics. In this work, integration of sub-nanoscale Ru species with NiFe LDH can dramatically enhance the adsorption energy of H* and improve their HER kinetics. Besides, benefitting from the desired potential-induced strategy, the Ru–NiFe LDH interfaces will convert to RuO2–NiFe(OOH)x interfaces to optimize the adsorption energy of OH* to meet the requirement of strengthening the OER performance. Strikingly, the Ru–NiFe LDH-F/NF sample (NF: Ni foam) shows an excellent OER and HER performance with an overpotential of 230.0 mV and 115.6 mV at a current density of 10 mA cm−2, respectively, as well as outstanding durability. The overall water splitting device was fabricated by using Ru/NiFe LDH-F/NF as both the HER and OER electrode with a potential of 1.53 V to achieve a current density of 10 mA cm−2. In addition, the theoretical calculations demonstrated that the Ru–NiFe LDH interfaces could optimize the adsorption energy of H* and OH*. This study provides a new insight into the development of highly efficient bifunctional electrocatalysts for water electrolysis.
- This article is part of the themed collection: Editor’s Choice: Single-atom and nanocluster catalysis


 Please wait while we load your content...
Please wait while we load your content...