Impact of cesium in methylammonium lead bromide perovskites: insights into the microstructures, stability and photophysical properties†
Abstract
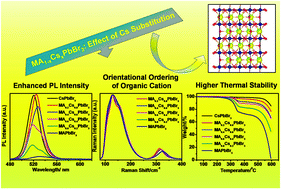
The thermal and moisture instabilities of pure organic lead halide perovskites are the foremost concerns towards the commercialization of perovskite solar cells, which can be avoided by introducing an inorganic cation, such as cesium ion (Cs+) at the A-site of the perovskite crystals. In this report, the impacts of substituted Cs+ cations on the inherent properties such as microstructures, morphology, and photophysics of pure methylammonium lead bromide (MAPbBr3) perovskites have been investigated. Successful formation of mixed MA1−xCsxPbBr3 phases (with 0 ≤ x ≤ 1.0) was predicted from the theoretically calculated tolerance factor, which was further supported by the appearance of sharp diffraction peaks in X-ray diffraction (XRD) patterns without any additional peaks in the whole composition range. Substitution of Cs+ ions brings significant lattice contraction in the parent MAPbBr3 crystal due to the ion size disparity in the ionic radii between MA+ and Cs+ ions. We examine the vibrational signatures of the Raman bands related to the organic MA+ and infer the nature of interactions between the organic moiety and the surrounding inorganic cage as a function of Cs concentration. Raman spectroscopic analysis reveals structural distortion due to the altered H-bonding interaction of the N+–H⋯Br− type between MA+ and the PbBr3− octahedral framework as a function of Cs content, which is responsible for the octahedral tilting in Cs substituted MAPbBr3. We also found hindered rotational motions of MA+ in the octahedral cage of mixed cationic systems, resulting in the orientational ordering of MA in the presence of Cs. These results certainly offer highly ordered mixed phase structures and promote superior thermal stability, as evident from the thermogravimetric analysis. The photoluminescence intensity becomes considerably enhanced at increased substitution levels, which highlights the capability of incorporated Cs+ cations in suppressing non-radiative recombination in a pure MA-based crystal, possibly related to the mitigation of trapping. The substitution of Cs+ with MAPbBr3 allows innovative strategies to improve the proficiency of tandem solar cells by modifying their structural and photophysical properties.


 Please wait while we load your content...
Please wait while we load your content...