Interfacing enzymes with silicon nanocrystals through the thiol–ene reaction†
Abstract
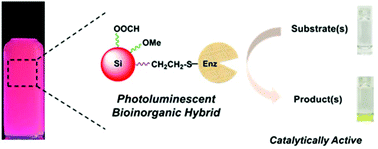
This study reports the preparation of functional bioinorganic hybrids, through application of the thiol–ene reaction, that exhibit catalytic activity and photoluminescent properties from enzymes and freestanding silicon nanocrystals. Thermal hydrosilylation of 1,7-octadiene and alkene-terminated poly(ethylene oxide)methyl ether with hydride-terminated silicon nanocrystals afforded nanocrystals functionalized with alkene residues and poly(ethylene oxide) moieties. These silicon nanocrystals were conjugated with representative enzymes through the photochemical thiol–ene reaction to afford bioinorganic hybrids that are dispersible and photostable in buffer, and that exhibit photoluminescence (λmax = 630 nm) and catalytic activity. They were characterized using Fourier transform infrared spectroscopy (FTIR), X-ray photoelectron spectroscopy (XPS), transmission electron microscopy (TEM), dynamic light scattering analysis (DLS), absorption spectroscopy, steady-state and time-resolved photoluminescence spectroscopy, and pertinent enzyme activity assays. The general derivatization approach presented for interfacing enzymes with biocompatible silicon nanocrystals has far reaching implications for many applications ranging from sensors to therapeutic agents. The bioinorganic hybrids presented herein have potential applications in the chemical detection of nitrophenyl esters and urea. They can also be employed in enzyme-based theranostics as they combine long-lived silicon nanocrystal photoluminescence with substrate-specific enzymatic activity.


 Please wait while we load your content...
Please wait while we load your content...