Single atom alloy catalyst for SO3 decomposition: enhancement of platinum catalyst's performance by Ag atom embedding†
Abstract
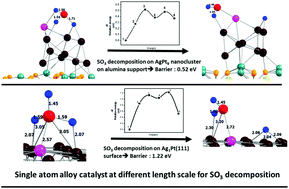
Recently, single atom alloy catalysts (SAA) have shown improved catalytic activity in numerous catalytic reactions. However, to date, single atom alloy (SAA) catalyst is not available for SO3 decomposition reaction, which is a key reactions in the hydrogen economy. Using state of the art density functional theory, we report a novel single Ag atom alloy Pt catalyst in the sub-nanometer length scale (AgPt9@Al2O3) showing superior catalytic behavior for SO3 decomposition. It was found that alloying the alumina-supported platinum nanocluster with a single Ag atom lowers the activation barrier for S–O bond breaking by more than 50% in comparison with the pristine platinum counterpart. Activation barrier for AgPt9@Al2O3 catalyst is 0.52 eV, which is the lowest of any platinum based catalyst reported so far. At variance with pure Pt10@Al2O3, which tries to detach from the support during decomposition reaction, single atom alloy (SAA) nanocluster AgPt9@Al2O3 enhances binding with support, thus strengthening sintering resistance. Notably, influence of single Ag atom is also observed at larger length scale, i.e., at Pt(111) slab, where single Ag atom substituted surface Ag1Pt(111) shows ∼30% reduction in activation barrier in contrast to a pristine surface. Single Ag atom works in bifunctional mode as it not only reduces the activation barrier, but also simultaneously weakly adsorbs the reaction product SO2, signifying relatively easier desorption and better recyclability. Deeper location of silver d-electrons and lesser electronegativity of silver is responsible for the better performance of single Ag atom alloyed Pt catalyst. We strongly believe that these remarkable results will open new avenues for future designing and fabrication of cost-effective catalysts for SO3 decomposition.
- This article is part of the themed collection: International Year of the Periodic Table : Single Atoms as Active Catalysts


 Please wait while we load your content...
Please wait while we load your content...