Well-dispersed Ni or MnO nanoparticles on mesoporous carbons: preparation via carbonization of bimetallic MOF-74s for highly reactive redox catalysts†
Abstract
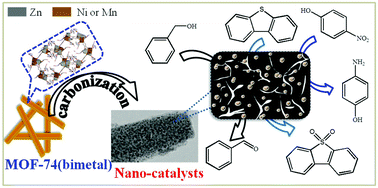
A series of metal–organic framework-74s (MOF-74s), composed of two different metallic species (Zn/Ni or Zn/Mn in various compositions), were synthesized, and Ni or MnO-doped carbonaceous materials were first prepared by pyrolysis of the MOFs under an inert environment for catalytic applications. These MOF-derived nanomaterials (MDNMs), obtained by pyrolysis of MOF-74s, were characterized thoroughly to understand their phase, porosity, particle size, dispersion, and composition. With increasing Zn content in the bimetallic MOF-74s, the porosity of the MDNMs increased but the size and content of Ni or MnO in the MDNMs decreased monotonously. One MDNM(75Zn25Mn), prepared from MOF-74(75%Zn/25%Mn), showed noticeably higher activity in the oxidation of benzyl alcohol as compared with not only the MDNM(xZnyMn)s but also MnOx-loaded carbon or loaded γ-alumina (or, MDNM(75Zn25Mn) showed ∼54 times turnover frequency (TOF) to that of MnO/activated carbon). MDNM(75Zn25Mn) was also effective in the oxidative removal of dibenzothiophene from a model fuel. Moreover, MDNM(75Zn25Ni), prepared from MOF-74(75%Zn/25%Ni), had the highest TOF in the reduction of 4-nitrophenol among various MDNM(xZnyNi)s. The highest activity of MDNM(75Zn25Mn) and MDNM(75Zn25Ni), even with the lowest Mn and Ni contents in the respective MDNMs, for oxidation and reduction in several cycles might be due to the well-dispersed MnO (and Ni) and high porosity with mesopores. Therefore, it can be suggested that pyrolysis of mixed-metal MOFs such as MOF-74s can be a facile way to obtain highly effective and recyclable heterogeneous catalysts, with well-dispersed active species in very small sizes, for various organic reactions.


 Please wait while we load your content...
Please wait while we load your content...