Intercalation of aromatic amine for the 2H–1T′ phase transition of MoS2 by experiments and calculations†
Abstract
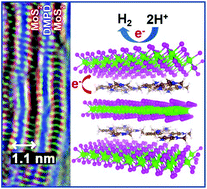
The novel properties of two-dimensional materials have motivated extensive studies focused on transition metal dichalcogenides (TMDs), which led to many interesting findings in recent years. Further advances in this area would require the development of effective methods for producing nanostructured TMDs with a controlled structure. Herein, we report unique MoS2 layered nanostructures intercalated with dimethyl-p-phenylenediamine (DMPD) with various concentrations, synthesized by a one-step hydrothermal reaction. The MoS2 layers possess a significantly expanded interlayer spacing. Remarkably, as the concentration of DMPD increases, the MoS2 preferentially adopts a unique metallic 1T′ (distorted 1T) phase. The intercalated MoS2 exhibits excellent catalytic performance in the hydrogen evolution reaction. First-principles calculations show that the phase transition from 2H to 1T′ phase occurs with increasing concentrations of DMPD, which can be accelerated by the S vacancies. A significant charge transfer from the DMPD molecules to MoS2 stabilizes the 1T′ over the 2H phase, driving the 2H–1T′ phase conversion. The DMPD and the S vacancies increased the carrier concentration, which leads to the enhanced catalytic performance. The present work illustrates how the phase control of TMDs can be effectively achieved by the intercalation of electron-donating molecules.


 Please wait while we load your content...
Please wait while we load your content...