Preparation of lactic acid- and glucose-responsive poly(ε-caprolactone)-b-poly(ethylene oxide) block copolymer micelles using phenylboronic ester as a sensitive block linkage†
Abstract
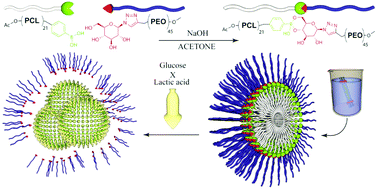
The present study describes the synthesis, self-assembly and responsiveness to glucose and lactic acid of biocompatible and biodegradable block copolymer micelles using phenylboronic ester as the linkage between hydrophobic poly(ε-caprolactone) (PCL) and hydrophilic poly(ethylene oxide) (PEO). The PCL block with pendant phenylboronic acid (PCLBA) was synthesized by combining ε-caprolactone (ε-CL) ring-opening polymerisation (ROP), using 4-hydroxymethyl(phenylboronic) acid pinacolate as the initiator, and pinacol deprotection. The glucose-terminated PEO (PEOGlc) was prepared by 1,3-dipolar, Cu(I)-catalysed, alkyne–azide cycloaddition of α-methoxy-ω-propargyl poly(ethylene oxide) and 1-azido-1-deoxy-D-glucopyranose. All new compounds were evaluated by 1H NMR spectroscopy and by SEC analysis. PCLBA and PEOGlc blocks were linked in NaOH acetone solution, which was indirectly confirmed by Alizarin Red S fluorescence and directly by 1H NMR spectroscopy. Dialysis against Milli-Q water induced the self-assembly of PCLBA-b-PEOGlc nanoparticles, which were characterised by static (SLS) and dynamic (DLS) light scattering and by cryogenic transmission electron microscopy (cryo-TEM). Furthermore, the microscopic properties of the charged interface between the hydrophobic PCLBA core and the hydrophilic PEOGlc shell were examined by electrophoretic light scattering (zeta potential) and by fluorescence spectroscopy using the fluorescent probe 5-(N-dodecanoyl)aminofluorescein (DAF) as a pH indicator. Subsequently, the nanoparticles were transferred to a phosphate buffer saline (PBS) solution supplemented with different concentrations of glucose to simulate the physiological conditions in blood or lactic acid to simulate acidic cytosolic or endosomal conditions in tumour cells. Adding a surplus of glucose or lactic acid, which competitively binds to PBA, removes the stabilising hydrophilic PEOGlc blocks, thereby triggering marked nanoparticle aggregation. However, the rate of aggregation induced by lactic acid is considerably faster than that induced by glucose, as confirmed by light scattering. Thus, this novel block copolymer may contribute to the field of selective, lactic acid- and/or glucose-responsive drug delivery vehicle design under both pathological and physiological conditions.


 Please wait while we load your content...
Please wait while we load your content...