Highly reversible and fast sodium storage boosted by improved interfacial and surface charge transfer derived from the synergistic effect of heterostructures and pseudocapacitance in SnO2-based anodes†
Abstract
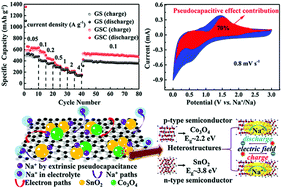
Sodium-ion batteries have attracted worldwide attention as potential alternatives for large scale stationary energy storage due to the rich reserves and low cost of sodium resources. However, the practical application of sodium-ion batteries is restricted by unsatisfying capacity and poor rate capability. Herein, a novel mechanism of improving both interfacial and surface charge transfer is proposed by fabricating a graphene oxide/SnO2/Co3O4 nanocomposite through a simple hydrothermal method. The formation of heterostructures between ultrafine SnO2 and Co3O4 could enhance the charge transfer of interfaces owing to the internal electric field. The pseudocapacitive effect, which is led by the high specific area and the existence of ultrafine nanoparticles, takes on a feature of fast faradaic surface charge-transfer. Benefiting from the synergistic advantages of the heterostructures and the pseudocapacitive effect, the as-prepared graphene oxide/SnO2/Co3O4 anode achieved a high reversible capacity of 461 mA h g−1 after 80 cycles at a current density of 0.1 A g−1. Additionally, at a high current density of 1 A g−1, a high reversible capacity of 241 mA h g−1 after 500 cycles is obtained. A full cell coupled by the as-prepared graphene oxide/SnO2/Co3O4 anode and the Na3V2(PO4)3 cathode was also constructed, which exhibited a reversible capacity of 310.3 mA h g−1 after 100 cycles at a current density of 1 A g−1. This method of improving both interfacial and surface charge transfer may pave the way for the development of high performance sodium-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...