Generating plasmonic heterostructures by cation exchange and redox reactions of covellite CuS nanocrystals with Au3+ ions†
Abstract
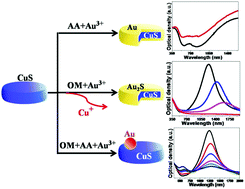
We demonstrate the fabrication of various types of heterostructures, including core–shells and dimers. This is achieved by reacting platelet-shaped covellite (CuS) nanocrystals (NCs) with Au3+ ions under various reaction conditions: the exposure of CuS NCs to Au3+ ions, in the presence or in the absence of ascorbic acid (AA), leads to the formation of CuS@Au core–shell nanostructures; the reaction of CuS NCs with Au3+ ions in the presence of oleylamine (OM) leads to the formation of CuS@Au2S; the presence of both OM and AA leads to the formation of Au/CuS dimers. Depending on which condition is chosen, either cation exchange (CE) between gold and copper ions is predominant (leading to amorphous Au2S) or the reduction of Au3+ leads to the nucleation of metallic Au domains (which are operated by the AA). In the heterostructures achieved by CE, the Au2S shell is almost entirely amorphous, and can be converted to polycrystalline upon electron beam irradiation. Finally, when both oleylamine and AA are present in the reaction environment, Au/CuS dimers are formed due to the reduction of Au3+ to metallic Au domains which nucleate on top of the CuS seeds. The experimental dual plasmonic bands of the CuS@Au core–shells and Au/CuS dimers are in agreement with the theoretical optical simulations. The procedures described here enable the synthesis of core–shell nanostructures with tunable localized surface plasmon resonances (LSPRs) in the near-infrared (NIR) region, and of plasmonic metal/semiconductor heterostructures with LSPRs in both the NIR and the visible regions.


 Please wait while we load your content...
Please wait while we load your content...