The combination of endolysosomal escape and basolateral stimulation to overcome the difficulties of “easy uptake hard transcytosis” of ligand-modified nanoparticles in oral drug delivery†
Abstract
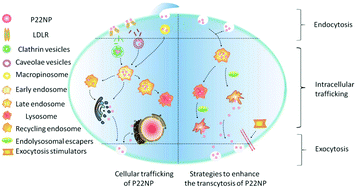
Ligand-modified nanoparticles (NPs) are an effective tool to increase the endocytosis efficiency of drugs, but these functionalized NPs face the drawback of “easy uptake hard transcytosis” in the oral delivery of proteins and peptides. Adversely, the resulting deficiency in transcytosis has not attracted much attention. Herein, NPs modified with the low-density lipoprotein receptor (LDLR) ligand NH2-C6-[cMPRLRGC]c-NH2, i.e., peptide-22 (P22NPs) were fabricated to investigate strategies related to the enhancement of transcytosis. By systematically studying the intracellular trafficking of NPs, it was found that reduced transcytosis might be associated with the entrapment of P22NPs in endosomes or lysosomes and limited basolateral exocytosis. On this basis, the prevention of the endolysosomal entrapment of NPs and the acceleration of basolateral exocytosis should be considered as strategies to enhance the transcytosis of NPs. By screening chemicals that could help the endosomal/lysosomal escape of chemicals related to LDLR-mediated transcytosis, it was shown that hemagglutinin-2 (HA2) and metformin had higher abilities to enhance the exocytosis of P22NPs. The transcytosis efficiencies of insulin loaded in P22NPs were also investigated, and a 3.2-fold increase in transcytosis was observed in comparison with free insulin. The transcytosis efficiencies of insulin could be further increased by the addition of metformin or HA2 (3.6-fold or 4.1-fold higher than that of free insulin). Inspiringly, the simultaneous addition of the abovementioned two chemicals led to the highest transcytosis efficiency of insulin, which was up to 5.1-fold higher than that of free insulin. These results demonstrated that endolysosomal entrapment and basolateral exocytosis are two of the most important limiting steps for the “easy uptake hard transcytosis” of orally administered ligand-modified NPs. Moreover, our work provides a new point of view for the design of novel oral drug delivery systems.


 Please wait while we load your content...
Please wait while we load your content...