Rational geometrical engineering of palladium sulfide multi-arm nanostructures as a superior bi-functional electrocatalyst†
Abstract
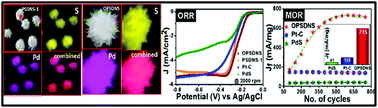
Geometrical tunability offers sharp edges and an open-armed structure accompanied with a high electrochemical active surface area to ensure the efficient and effective utilization of materials by exposing the electrochemical active sites for facile accessibility of reactant species. Herein, we report a one-step, single-pot, surfactant-free, electroless, and economic route to synthesize palladium sulfide nanostructures with different geometries at mild temperatures and their catalytic properties towards the oxygen reduction reaction (ORR) and methanol electro-oxidation (MOR). For ORR, the positive on-set, half wave potentials, smaller Tafel slope, high electrochemical active surface area, large roughness factor, and better cyclic stability of the proposed nanostructures as compared to those of the commercial state-of-the-art Pt–C/PdS catalysts suggest their superiority in an alkaline medium. In addition, high mass activity (Jf ∼ 715 mA mg−1), in comparison with that of the commercial state-of-the-art Pt–C/PdS catalysts (Jf ∼ 138/41 mA mg−1, respectively), and high Jf/Jb (1.52) along with the superior operational stability of the multi-arm palladium sulfide nanostructures towards MOR advocates the bi-functional behavior of the catalyst and its potential as a promising Pt-free anode/cathode electrocatalyst in fuel cells.


 Please wait while we load your content...
Please wait while we load your content...