Nanostructuration of ionic liquids: impact on the cation mobility. A multi-scale study†
Abstract
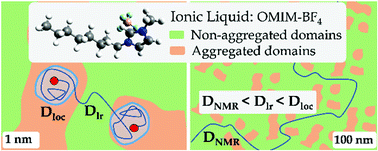
When probed at the macroscopic scale, Ionic Liquids (ILs) behave as highly dissociated (i.e. strong) electrolytes while, at the molecular scale, they show clear characteristics of weak ionic solutions. The multi-scale analysis we report in this paper reconciles these apparently at odds behaviors. We investigate by quasi-elastic neutron scattering (QENS) and neutron spin-echo (NSE), the nanometer/nanosecond dynamics of OMIM-BF4, an imidazolium-based IL showing strong nanostructuration. We also probe the same IL on the microscopic (μm and ms) scale by pulsed field gradient NMR. To interpret the neutron data, we introduce a new physical model to account for the dynamics of the side-chains and for the diffusion of the whole molecule. This model describes the observables over the whole and unprecedented investigated spatial ([0.15–1.65] Å−1) and time ([0.5–2000] ps) ranges. We arrive at a coherent and unified structural/dynamical description of the local cation dynamics: a localized motion within the IL nanometric domains is combined with a genuine long-range translational motion. The QENS, NSE and NMR experiments describe the same long-range translational process, but probed at different scales. The associated diffusion coefficients are more than one order of magnitude different. We show how this apparent discrepancy is a manifestation of the IL nanostructuration.


 Please wait while we load your content...
Please wait while we load your content...