CDC20siRNA and paclitaxel co-loaded nanometric liposomes of a nipecotic acid-derived cationic amphiphile inhibit xenografted neuroblastoma†
Abstract
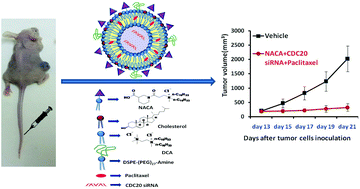
Despite significant recent progress in the area of translational genomics of neuroblastoma, the overall survival rates for children with high-risk NB continue to be not more than 5 years due to tumor relapse and/or drug-resistant tumors. Herein we report on the development of a neuroblastoma targeting nanometric (130–150 nm) circulation stable liposomal system prepared from a novel nipecotic acid-derived cationic amphiphile (NACA). The size ranges of liposomes (130–150 nm) were confirmed by both dynamic light scattering and transmission electron microscopy. The findings in the gel electrophoresis assay revealed that siRNAs encapsulated within the liposomes of NACA (with 90% entrapment efficiency) are protected from attack by RNase. Cellular uptake experiments using FAM-siRNA loaded liposomes of NACA showed the liposomal entry in human neuroblastoma cells (IMR-32) to be mediated via the GABAA receptor. CDC20siRNA-loaded liposomes of NACA caused significantly higher CDC20 gene silencing efficiency in IMR-32 cells compared to CDC20 gene knockdown efficiency mediated by CDC20siRNA-loaded control non-targeting liposomes (NTL). The findings in the annexin-V binding based flow cytometric apoptosis assay and MTT-based cellular cytotoxicity assay support the notion that pronounced (80%) neuroblastoma cell death upon treatment with CDC20siRNA & PTX co-loaded liposomes of NACA presumably originates from enhanced apoptosis of cells. Importantly, intravenously administered CDC20siRNA & PTX co-loaded liposomes of NACA significantly inhibited growth of xenografted human neuroblastoma in athymic nude mice. The presently disclosed strategy of co-delivering potent anticancer siRNA and small molecule chemotherapeutics using liposomes of NACA opens a new door for combating the dreaded disease of neuroblastoma.


 Please wait while we load your content...
Please wait while we load your content...