A novel approach for the synthesis of ultrathin silica-coated iron oxide nanocubes decorated with silver nanodots (Fe3O4/SiO2/Ag) and their superior catalytic reduction of 4-nitroaniline†
Abstract
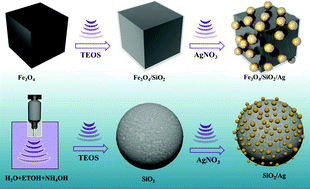
A novel sonochemical approach was developed for the synthesis of different core/shell structures of Fe3O4/SiO2/Ag nanocubes and SiO2/Ag nanospheres. The total reaction time of the three sonochemical steps for the synthesis of Fe3O4/SiO2/Ag nanocubes is shorter than that of the previously reported methods. A proposed reaction mechanism for the sonochemical functionalization of the silica and the silver on the surface of magnetic nanocubes was discussed in detail. Transmission electron microscopy revealed that the surface of Fe3O4/SiO2 nanocubes was decorated with small Ag nanoparticles of approximately 10–20 nm in size, and the energy dispersive spectroscopy mapping analysis confirmed the morphology of the structure. Additionally, X-ray diffraction data were used to confirm the formation of both phases of a cubic inverse spinel structure for Fe3O4 and bcc structures for Ag in the core/shell structure of the Fe3O4/SiO2/Ag nanocubes. The as-synthesized Fe3O4/SiO2/Ag nanocubes showed a high efficiency in the catalytic reduction reaction of 4-nitroaniline to 4-phenylenediamine and a better performance than both Ag and SiO2/Ag nanoparticles. The grafted silver catalyst was recycled and reused at least fifteen times without a significant loss of catalytic efficiency.

 Please wait while we load your content...
Please wait while we load your content...