Complementary mass spectrometric techniques for the quantification of the protein corona: a case study on gold nanoparticles and human serum proteins†
Abstract
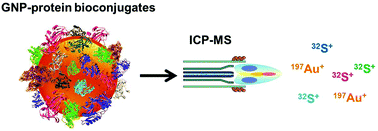
Once nanoparticles enter a biological system, it is known that their surface is instantly covered by the biomolecules present with preference to proteins. This protein corona has been a subject of numerous studies in order to reveal its composition. Besides that, growing interest exists in its quantitative determination in order to gain a deeper insight into the nature of these nanoparticle–protein bioconjugates. Only a few analytical methods are available nowadays, so the aim of this study is to provide a reliable and alternative methodology for the quantification of the protein corona. The suggested approach is based on the assumption that the total protein content within the corona can be correlated to its sulfur concentration due to the presence of cysteine and methionine as sulfur-containing amino acids. Once the most abundant proteins had been identified with the use of gel electrophoresis with subsequent peptide analysis by electrospray ionization-tandem mass spectrometry (ESI-MS/MS), the isolated nanoparticle–protein conjugates were subjected to total analysis of sulfur and the corresponding metal being present in the nanoparticles by inductively coupled plasma-mass spectrometry (ICP-MS). The concept is exemplarily demonstrated on citrate-stabilized gold nanoparticles (GNPs) incubated with human serum. Two different purification procedures were tested in order to isolate the sought bioconjugates. 26 most abundant proteins could be identified and an average of approximately 40 S atoms per protein was calculated and used for further studies. ICP-MS analyses of S/Au ratios served for the quantification of the protein corona revealing an absolute number of proteins bound to the incubated GNPs. Two main results could be obtained for this specific system under the chosen experimental conditions: the number of proteins per GNP decreased with their size from 10 nm to 60 nm and the obtained values suggested that the protein corona in this specific case was theoretically formed either as a monolayer (60 nm GNPs) or as a multilayer (5–7 protein layers per 10 nm GNP). Studies with bovine serum albumin (BSA) as the model protein showed similar results.

 Please wait while we load your content...
Please wait while we load your content...