Iron oxide nanoparticles immobilized to mesoporous NH2-SiO2 spheres by sulfonic acid functionalization as highly efficient catalysts†
Abstract
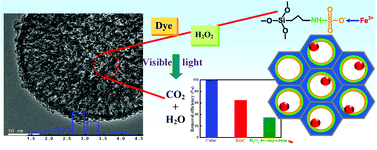
A novel SiO2 nanosphere was synthesized by the post-synthetic grafting of sulfonic acid groups on to anionic-surfactant-templated mesoporous NH2-silica (AMAS). This one-pot post-functionalization strategy allowed more metal ions to be homogeneously anchored into the channel of the meso-SiO2 nanosphere. After hydrothermal and calcination treatment, the in situ growth of α-Fe2O3 on sulfonic acid-functionalized mesoporous NH2–SiO2 (SA-AMAS) exhibited much higher activity in the visible-light assisted Fenton reaction at neutral pH than that for AMAS or meso-SiO2 nanospheres. By analysis, the grafted sulfonic acid group can not only enhance the acid strength of the catalyst, but can also bring more orbital-overlapping between the active sites (FeII and FeIII) and the surface peroxide species, to facilitate the decomposition of H2O2 to hydroxyl radical. The present results provide opportunities for developing heterogeneous catalysts with high-performance in the field of green chemistry and environmental remediation.

 Please wait while we load your content...
Please wait while we load your content...