Heterostructured Ag3PO4/AgBr/Ag plasmonic photocatalyst with enhanced photocatalytic activity and stability under visible light†
Abstract
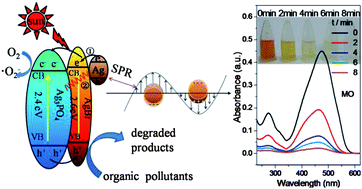
A heterostructured Ag3PO4/AgBr/Ag plasmonic photocatalyst was prepared by a rational in situ ion exchange reaction between Ag3PO4 micro-cubes and Br− in aqueous solution followed by photoreduction. The photocatalytic activities of obtained photocatalysts were measured by the degradation of methyl orange (MO) and methylene blue (MB) under visible light irradiation (λ ≥ 400 nm). Compared to AgBr/Ag, Ag3PO4/AgBr heterocrystals and pure Ag3PO4 crystals, the heterostructured Ag3PO4/AgBr/Ag plasmonic photocatalysts exhibit much higher photocatalytic activity and stability. This enhanced photocatalytic activity suggests that the synergetic effects of the heterostructured Ag3PO4/AgBr/Ag and the strong SPR of Ag NPs on the surface result in the high efficiencies of the photocatalytic activity and the improved stability. With the assistance of Ag3PO4/AgBr/Ag heterostructures, only 8 min and 12 min are taken to completely decompose MO and MB molecules under visible-light irradiation, respectively. Furthermore, the photodegradation rate does not show an obvious decrease during ten successive cycles, indicating that our heterostructured Ag3PO4/AgBr/Ag plasmonic photocatalysts are extremely stable under visible-light irradiation.

 Please wait while we load your content...
Please wait while we load your content...