The influence of triphenylamine as a donor group on Zn–porphyrin for dye sensitized solar cell applications
Abstract
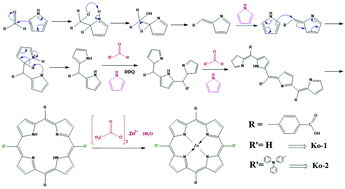
Two donor–π–acceptor type dyes, Zn[10,20-(4-carboxyphenyl)porphyrin] (Ko-1) and Zn[5,15-diphenylaminophenyl-10,20-(4-carboxyphenyl)porphyrin] (Ko-2), with two different donor units were designed and synthesized for solar cell applications. The Ko-2 dye contains the triphenylamine group as an electron donor group, the porphyrin unit acts as a π-bridge, and the carboxylic acid group acts as an anchoring group (electron acceptor unit). The Ko-2 dye has a red-shifted absorption maxima owing to the introduction of the dimethylaminophenyl moiety at the meso position of the porphyrin ring. The highly conjugated dimethylaminophenyl group efficiently donates electrons and the electronic interaction between the porphyrin and dimethylaminophenyl unit is better compared to hydrogen. A dye sensitized solar cell (DSSC) was made using commercial P25 TiO2 material as a photoanode, the Zn–porphyrin derivatives as sensitizers, I−/I3− as an electrolyte, and platinum (Pt) as the counter electrode. The molar extinction coefficient, highest occupied molecular orbital (HOMO), and lowest unoccupied molecular orbital (LUMO) values of the Ko-2 dye are 1.73 × 105 M−1 cm−1, −5.82 eV, and −3.35 eV, respectively, which are more suitable for DSSC applications than those of the Ko-1 dye, which were 1.31 × 105 M−1 cm−1, −6.16 eV, and −3.60 eV, respectively. The solar cell performance of the Ko-2-based DSSC reached 3.3% efficiency with an open circuit voltage (Voc) of 0.68 V, a short circuit photocurrent density (Jsc) of 9.69 mA cm−2, and a fill factor (FF) of 0.49; the Ko-1-based DSSC reached 1.90% efficiency, a Voc of 0.67 V, a Jsc of 5.51 mA cm−2, and a FF of 0.54 under AM 1.5 G irradiation.


 Please wait while we load your content...
Please wait while we load your content...