Theoretical insights into the diffusion mechanism of alkali ions in Ruddlesden–Popper antiperovskites†
Abstract
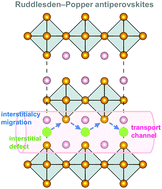
Antiperovskites exhibit promising potential in the application of all-solid-state batteries due to their high ionic conductivity. A comprehensive understanding of the diffusion mechanism of alkali ions in antiperovskites is significant to explore novel antiperovskite electrolyte materials. In this work, we report the migration mechanism of alkali ions in Ruddlesden–Popper (RP) antiperovskites A4OX2 (A = Li, Na; X = Cl, Br, I) through first-principles calculations. Migration mechanisms of different vacancies and interstitials are simulated for these RP antiperovskites. The alkali interstitials are predicted to have lower migration barriers than vacancies, and play a significant role in the ionic conduction of RP antiperovskites. Therefore, the increase of the content of alkali interstitials is a reasonable approach to further improve the ionic conductivity of RP antiperovskites. Our results can serve as a useful guide for developing novel antiperovskite electrolytes of all-solid-state batteries.


 Please wait while we load your content...
Please wait while we load your content...