The one-pot synthesis of porous Ni0.85Se nanospheres on graphene as an efficient and durable electrocatalyst for overall water splitting†
Abstract
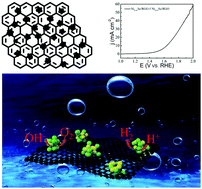
The design and preparation of highly efficient and durable non-noble metal electrocatalysts for overall water splitting is pivotal for actualizing large-scale water splitting via electrolysis. Herein, porous Ni0.85Se nanospheres anchored on reduced graphene oxide (RGO) (Ni0.85Se/RGO) were synthesized via a one-step hydrothermal method. Following explorations of the hydrothermal temperature and reaction time, the optimized Ni0.85Se/RGO exhibited extraordinary oxygen evolution reaction (OER) and hydrogen evolution reaction (HER) performance in 1.0 M KOH solution, providing an overpotential as small as 320 mV at a current density of 30 mA cm−2 and a low Tafel slope of 91 mV dec−1 for the OER, and an overpotential of 169 mV at a current density of 10 mA cm−2 and a low Tafel slope of 65 mV dec−1 for the HER. A current density of 10 mA cm−2 at 1.64 V and high stability can be obtained upon using Ni0.85Se/RGO as both the cathode and anode for overall water splitting under alkaline conditions; this was attributed to the merits of both the porous structure of Ni0.85Se and the high conductivity of RGO. This work provides a meaningful exploration of the synthesis of 3d-transition-metal-chalcogenide- and carbon-based composite electrocatalysts via a hydrothermal reaction for water splitting, and provides a general method.


 Please wait while we load your content...
Please wait while we load your content...