The preparation of NiO/Ni–N/C nanocomposites and its electrocatalytic performance for methanol oxidation reaction†
Abstract
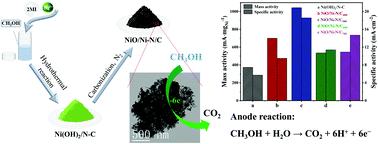
Direct methanol fuel cells (DMFCs) have been extensively studied for developing a new non-precious metal catalyst to replace commercial platinum catalysts, due to their expensive cost and susceptibility to CO poisoning. In this work, nanocomposites of nickel oxide/nickel supported by N-doped carbon (NiO/Ni–N/C) were prepared by using a hydrothermal method and further carbonization at different temperatures. The surface morphology and composition of the NiO/Ni–N/C nanocomposites were characterized by using X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM) and X-ray photoelectron spectroscopy (XPS). As observed through electrochemical experiments, the NiO/Ni–N/C nanocomposite modified glassy carbon electrode (NiO/Ni–N/C/GCE) exhibits high electrocatalytic activity for the methanol oxidation reaction (MOR) in alkaline solution. Among the nanocomposites, NiO/Ni–N/C carbonized at 500 °C (NiO/Ni–N/C500) displays the highest mass activity (1043 mA mgNi−1) at 0.73 V (vs. Ag/AgCl), which is much higher than that of Ni(OH)2/N–C and NiO/Ni–N/C carbonized at other temperatures (400, 600, and 700 °C). In addition, the catalytic activity of NiO/Ni–N/C500 is 2.12 times that of the commercial Pt/C. NiO/Ni–N/C500/GCE also possesses fast kinetics, excellent stability and CO tolerance. Therefore, the NiO/Ni–N/C500 nanocomposites have great application potential as MOR catalysts.


 Please wait while we load your content...
Please wait while we load your content...