A cellulose dissolution and encapsulation strategy to prepare carbon nanospheres with ultra-small size and high nitrogen content for the oxygen reduction reaction†
Abstract
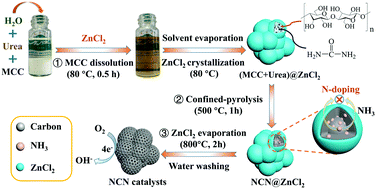
We have devised a feasible approach to prepare nitrogen-doped carbon nanospheres (NCNs) with ultrasmall size and high N-doping amount via the dissolution and space-confined pyrolysis of cellulose using ZnCl2. The results indicated that pyrolysis, gasification, and nitrogen doping of the cellulose occurred in a fully-sealed nanoreactor (inner diameter ≈ 11 nm). The formation of an autogenic pressure inside the nanoreactor played an important role in improving the heteroatomic equilibrium conditions, thereby increasing the nitrogen content (increased to ca. 2 times) and biochar yield (increased by ca. 56.6%). The nitrogen content, nitrogen type and equivalence ratio, as well as the specific surface area, were easily regulated by varying the amount of ZnCl2. The resulting NCN-7 exhibited a high oxygen reduction reaction (ORR) activity with a half-wave potential (E1/2) of 0.87 V (versus the reversible hydrogen electrode, RHE) that surpassed those of most reported biomass-derived catalysts, even those of the commercial Pt-based catalysts (0.86 V). This remarkably improved catalytic activity was attributed to the high total nitrogen content (8.56 at%), pyridinic N concentration (3.06 at%), and large specific surface area (1655.0 m2 g−1) of the catalyst. This work proposes a universal method for the conversion of abundant renewable biomass (including cellulose, chitosan and cotton-stalks) into advanced nanoelectrocatalysts by the dissolution and space-confined pyrolysis strategy.


 Please wait while we load your content...
Please wait while we load your content...