Synergistic effect of cobalt, nitrogen-codoped hollow carbon sphere hosts for high performance lithium sulfur batteries†
Abstract
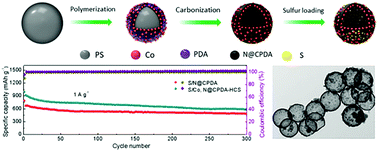
Lithium–sulfur (Li–S) batteries are considered to be one of the most promising candidates for the next generation of rechargeable batteries due to their high theoretical energy density and low cost. However, the practical application of Li–S batteries is seriously hindered by the dissolution of polysulfide and the intrinsic insulation of sulfur. Herein, we demonstrate a novel strategy to address these two problems by designing and synthesizing a cobalt, nitrogen-codoped hollow carbon sphere (Co, N@CPDA-HCS) with a highly conductive carbon shell to accommodate the sulfur. In this unique material architecture, each component synergistically serves a specific purpose: the highly conductive shell carbon can provide fast electron conduction paths and structural stability; the superfine Co nanoparticles uniformly embedded in the carbon shell can accelerate the conversion kinetics of the polysulfides by strong chemical interaction; moreover, the hollow structure provides a buffer space for the volume change of sulfur during charging and discharging, thus leading to a good cycle stability of sulfur cathodes. Co, N@CPDA-HCS exhibits balanced high electrochemical performance with respect to specific capacity, rate capability, and cycling stability. Specifically, S/Co, N@CPDA-HCS presents a high initial gravimetric capacity of 1365 mA h g−1 at 0.1 A g−1. The discharge capacity of 588 mA h g−1 still remained at the current density of 1 A g−1 after 300 cycles.


 Please wait while we load your content...
Please wait while we load your content...