Effects of a Ni cocatalyst on the photocatalytic hydrogen evolution reaction of anatase TiO2 by first-principles calculations†
Abstract
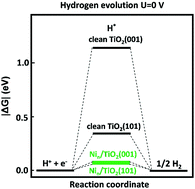
TiO2 is a promising photocatalyst for overall water splitting, and its low efficiency for the hydrogen evolution reaction (HER) can be improved by loading proper cocatalysts. Accordingly, to explore the working mechanism of Ni/TiO2 photocatalysts in the HER, we performed in-depth first-principles calculations. It was found that it is more difficult for the Nin cluster to undergo aggregation on the TiO2(001) surface than the TiO2(101) surface. The structural difference between these two surfaces determines the above-mentioned property. The adsorption of the Nin cluster on the TiO2 surface is beneficial for the separation of photogenerated charges by introducing mid-gap states into the band gap and elevating the Fermi level to a higher energy region, which is favorable for the HER. An electrochemical computational method was applied to investigate the process of producing hydrogen. Our results indicate that the active site of HER in Nin/TiO2 composites is the O2c atoms binding or near to the Nin cluster. Thus, loading Nin clusters remarkably decreases the Gibbs free energy of the HER compared with clean surfaces. Our study reveals the growth behavior of the Ni cocatalyst on the anatase TiO2 surfaces and presents a reasonable explanation for the experimental improvement in the photocatalytic activity of TiO2 for HER upon introducing Ni cocatalysts.


 Please wait while we load your content...
Please wait while we load your content...