A combined experimental and theoretical study of bimetallic bis- and tris-homocubane analogues†
Abstract
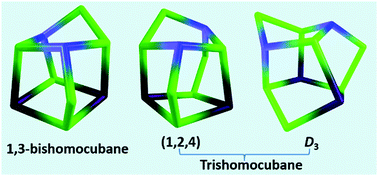
Synthesis of various metal-incorporated bis- and tris-homocubane analogues has been reported. Room temperature reactions of [Cp*MCl2]2 (Cp* = η5-C5Me5, M = Ir or Rh) with chalcogenated borohydride reagents, Li[BH2E3] (E = S or Se), yielded a series of bimetallic bis- and tris-homocubane derivatives (1–7). The bishomocubane analogues belong to the 1,3-bishomocubane family with the general formula [(Cp*M)2(μ-E)2(μ3-E)4(μ3-BH)2] (1: M = Ir, E = S; 2: M = Ir, E = Se; 5: M = Rh, E = S and 6: M = Rh, E = Se), and [(Cp*Ir)2(μ-S)2(μ3-S)4(μ-BH2)2], 3, can be described as an unusual bishomocubane having two (μ-BH2) units with a missing-bond. In addition to these bishomocubanes, two trishomocubane derivatives [(Cp*M)2(μ-E)3(μ3-E)4(μ3-BH)2] (4: M = Ir, E = S and 7: M = Rh, E = Se) were isolated from the above reactions. Trishomocubane 4 adopts a 1,2,4-trishomocubyl structure, whereas 7 is a D3-trishomocubyl analogue. In a similar fashion, thermolysis of [Cp*CoCl]2 with Li[BH2E3] (E = S or Se) led to the formation of Co-1,3-bishomocubane analogues, [(Cp*Co)2(μ-E)2(μ3-E)4(μ3-BH)2] (8: E = S and 9: E = Se). All the compounds were characterized by multinuclear NMR and IR spectroscopies and mass spectrometric analysis. The core geometries of 1–4 and 8 were unequivocally established by single-crystal X-ray diffraction studies. Density functional theory (DFT) computations further demonstrated that metals and chalcogen atoms play an important role in determining the thermodynamic stability of the bis- and tris-homocubane species.
- This article is part of the themed collection: Selenium & Tellurium chemistry at the beginning of the 3rd millennium: a celebration of ICCST


 Please wait while we load your content...
Please wait while we load your content...