Solvothermal synthesis of CuCoO2 nanoplates using zeolitic imidazolate framework-67 (ZIF-67) as a co-derived precursor
Abstract
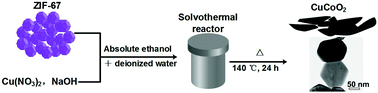
Herein, we reported for the first time that copper-based delafossite CuCoO2 nanoplates (ca. 150 nm) were successfully prepared by the solvothermal method at 140 °C for 24 hours using zeolitic imidazolate framework-67 (ZIF-67) as the Co precursor. The crystalline size and morphology could be easily controlled by simply modulating the amount of NaOH mineralizer added and reactant precursors. The crystal composition, morphology and crystal structures of the samples were systematically analyzed by powder X-ray diffraction (PXRD), field-emission scanning electron microscopy (FESEM), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS) and inductively coupled plasma-optical emission spectrometry (ICP-OES). Moreover, the chemical states, Brunauer–Emmett–Teller (BET) surface area and thermal stability of the CuCoO2 samples (ca. 150 nm) were systemically characterized. We believe that our synthesis strategy will open a new route for the preparation and application of CuCoO2, which involves combining the advantages of ZIF-67 and various components or structures. Furthermore, the resulting CuCoO2 nanoplates can serve as a potential candidate for a cost-effective and active electrode material.


 Please wait while we load your content...
Please wait while we load your content...