Coupling of fluoroborylene ligands in manganese carbonyl chemistry to give a difluorodiborene ligand†
Abstract
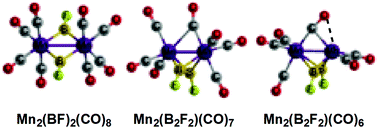
The geometries and energetics of the binuclear fluoroborylene manganese carbonyls Mn2(BF)2(CO)n (n = 8, 7, 6) have been investigated using density functional theory. Coupling of two fluoroborylene ligands to give a bridging difluorodiborene μ-B2F2 ligand with a relatively long ∼1.9 Å B–B bond has been observed in two of the three lowest energy Mn2(BF)2(CO)7 structures as well as the lowest energy Mn2(BF)2(CO)6 structure. Higher energy Mn2(BF)2(CO)6 structures have two separate bridging BF groups and short Mn![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Mn distances of ∼2.3 Å suggesting the formal triple bond required by the 18-electron rule.
Mn distances of ∼2.3 Å suggesting the formal triple bond required by the 18-electron rule.


 Please wait while we load your content...
Please wait while we load your content...