A fluorescein-based chemosensor for “turn-on” detection of Hg2+ and the resultant complex as a fluorescent sensor for S2− in semi-aqueous medium with cell-imaging application: experimental and computational studies†
Abstract
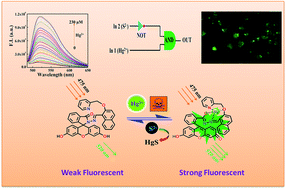
A fluorescein hydrazone based conjugate (H2L3) was synthesized by coupling fluorescein hydrazide (L1) with 2-(pyridin-2-ylmethoxy)-naphthalene-1-carbaldehyde (L2). H2L3 was well characterized by several spectroscopic methods such as IR, 1H, 13C-NMR and ESI-MS. The probe H2L3 exhibited high selectivity and high sensitivity towards toxic Hg2+ ions in semi-aqueous medium (pH 7.2, 10 mM HEPES) over other metal ions. The significant enhancement in fluorescence emission centered at 520 nm was attributed to the Hg2+-induced ring opening of the spirolactam moiety in the fluorescein structure. The 1 : 1 binding of H2L3 to Hg2+ was established by Job's method and confirmed by ESI-MS+ (m/z) studies and the binding constant was calculated as (0.43 ± 0.04) × 104 M−1 with a detection limit of 1.24 μM. Then again, the [Hg(HL3)]+ ensemble could be utilized as a reversible fluorescent sensor for S2−. On addition of S2− to the [Hg(HL3)]+ complex, the fluorescence intensity was totally quenched because Hg2+ in the complex was grabbed by S2− because of the stronger binding force between Hg2+ and S2−. The tentative coordination environment in the [Hg(HL3)]+ complex was established by DFT studies. The fluorescence “OFF–ON–OFF” mode of H2L3 was examined in the presence of Hg2+ and S2− and finds applications in devices with logic gate functions. H2L3 also exhibits bio-compatibility and negligible cytotoxicity and is suitable for fluorescence cell imaging of Hg2+ ions in live HepG2 cells.


 Please wait while we load your content...
Please wait while we load your content...