A simple route to surface functionalization of graphene nanosheets by benzoic acid and its application toward Pb(ii) sensing
Abstract
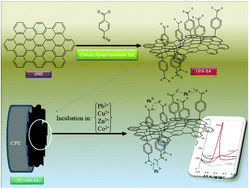
Herein, graphene nanosheets (GNS) were synthesized using a green and easy method by the reduction of graphene oxide with glucose. Then the surface of the GNS was functionalized with benzoic acid via a diazonium grafting route to produce benzoic acid functionalized graphene (GNS-BA). The prepared samples were characterized by atomic force microscopy (AFM), transmission electron microscopy (TEM), X-ray diffraction (XRD), and Fourier transfer infrared (FTIR) spectroscopy. A carbon paste electrode (CPE) was modified with GNS-BA (CPE/GNS-BA). The prepared modified electrode was electrochemically characterized by using [Fe(CN)6]3−/4− as a redox probe. Stripping voltammetry responses of the pre-concentrated ions on top of the modified electrode were used to evaluate the sensing ability of the CPE/GNS-BA electrode toward cations. The CPE/GNS-BA electrode showed that immobilized BA improved the accumulation performance for Pb(II) ion and parameters affecting the performance, such as pre-concentration time, solution pH, and ion concentration, were optimized for the stripping voltammetry response of the electrode. Subsequent to instrumental optimization, a calibration curve from 1.0 × 10−9 to 1.0 × 10−3 M Pb(II), r2 = 0.998 (±0.002), with a sensitivity of 0.265 (±0.003) mA cm−2, DL of 1.5 × 10−10 M Pb(II), and RSD of 5.7% was observed. This work provides a simple strategy for the construction of new ion adsorbents and sensors based on the molecular functionalization of graphene nanosheets via a diazonium grafting route.


 Please wait while we load your content...
Please wait while we load your content...