Design, synthesis, and characterization of an Fe(ii)-polymer of a redox non-innocent, heteroatomic, polydentate Schiff's base ligand: negative differential resistance and memory behaviour†
Abstract
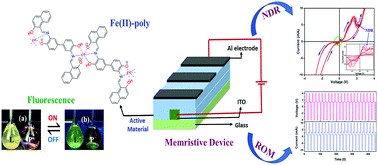
In the present work, the unique negative differential resistance (NDR) and memory effect of an organic–metallic hybrid polymer based on the self-assembly of Fe(II)-ions preceded by the synthesis of a newly designed multidentate Schiff's base ligand has been reported. The polymerization process was closely monitored in an absorbance study using a UV-vis spectrophotomer. The presence of sharp isosbestic points and saturation of absorbance at 1 : 1 ligand to metal concentration confirmed the stepwise growth of the polymer. A model monomer Fe-complex was also developed based on the model ligand, with the aim of understanding the properties of the polymer in a better way by comparative study but surprisingly both ligand and model ligand coordinate in a different fashion with the same Fe(II)-metal ions under similar reaction conditions, though both of them have identical binding sites. It has been found that the monomer model complex is paramagnetic while the metallopolymer is diamagnetic in nature. FESEM images of thin films of the polymer revealed the presence of homogeneously distributed interparticulate mesopores which was further confirmed by surface area analysis. A long single strand of the polymer was found on the HOPG surface by AFM study. The cyclic voltammetry study indicated that both conjugated ligand and metallopolymers are electrochemically active. The bistable memory behaviour of the device fabricated on ITO has shown a negative differential resistance effect along with good RAM and ROM behaviour, showing the potential of this novel polymer as memristor.


 Please wait while we load your content...
Please wait while we load your content...