Enhanced Z-scheme photocatalytic activity of a π-conjugated heterojunction: MIL-53(Fe)/Ag/g-C3N4†
Abstract
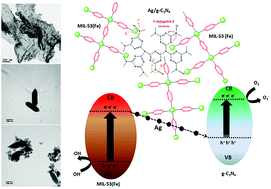
Remarkably competent visible light induced MIL-53(Fe)/Ag/g-C3N4 photocatalysts were fabricated by a facile ultrasonic technique. MIL-53(Fe)/Ag/g-C3N4 exhibits a Z-scheme charge transfer system, which demonstrates excellent photocatalytic activity in contrast to that of pristine catalysts upon exposure to visible-light (λ > 400 nm). Clioquinol was chosen as the target pollutant. Photoluminescence analysis showed that the utilization of varying weight ratios of Ag/g-C3N4 to MIL-53(Fe) improved the segregation and transfer rate of photo-generated excitons, thus resulting in the formation of a Z-scheme charge transfer system. Time dependent density functional theory calculations were also employed for both MIL-53(Fe) and g-C3N4 in order to predict the interactions of the π-conjugated system in the heterojunction. The morphology, crystallinity and opto-electronic properties of the material were reported in order to analyze the photoelectric behavior of the photocatalyst. Cluster charges for both MIL-53(Fe) and g-C3N4 and their interactions were theoretically calculated in order to predict the charge distribution between the clusters. Based on the results obtained from the theoretical and experimental calculations, a mechanism of photodegradation for the Z-scheme system was proposed.


 Please wait while we load your content...
Please wait while we load your content...