N-Fluoro functionalization of heterocyclic azoles: a new strategy towards insensitive high energy density materials†
Abstract
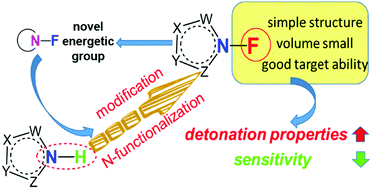
As a kind of dangerous material, energetic materials have the characteristics of being thermally unstable and easily combustible or explode upon exposure to external stimuli. The design and synthesis of novel insensitive energetic materials with high performance, therefore, have attracted continuing interest from researchers around the world. Hydroxylammonium salts were reported to be the most promising energetic salts due to their high densities and good oxygen balance. Energetic compounds consisting of N–F bonds are also expected to be denser and have better oxygen balance than their precursors. Herein, three series of new N–F compounds were designed and their physicochemical properties such as densities, heats of formation, detonation properties, and impact sensitivities, were evaluated by using volume-based thermodynamic calculations and density functional theory. Their properties were also compared with those of the corresponding hydroxylammonium salts. Among the investigated compounds, tris(4,5-dinitro-imidazol-2-yl)amine (TA2) and tris(5-nitro-1,2,4-triazol-3-yl)amine (TB2) displayed excellent integrated performance, that is, high density (>1.93 g cm−3), detonation velocity (>9000 m s−1), and detonation pressure (>37.0 GPa). These results are expected to facilitate the experimental synthesis of a new-generation of N–F based high explosives.


 Please wait while we load your content...
Please wait while we load your content...