Designing a biomimetic catalyst for phenoxazinone synthase activity using a mesoporous Schiff base copper complex with a novel double-helix morphology
Abstract
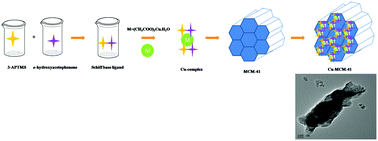
The condensation reaction of 3-aminopropyltrimethoxysilane with o-hydroxyacetophenone and subsequent complexation with Cu(CH3COO)2·H2O afforded a Schiff base copper complex with a silane moiety. Covalent functionalisation of MCM-41 with the Schiff base copper complex yielded a heterogenised copper complex which mimics the “CuN2O2” coordination site that resembles the galactose oxidase enzyme. The synthesized materials were characterized by UV-DRS, Fourier transform infrared spectroscopy (FTIR), thermogravimetric analysis (TGA), scanning electron microscopy (SEM) and energy dispersive X-ray analysis (EDAX) studies along with high resolution transmission electron microscopy (HR-TEM) to confirm successful functionalisation. Low-angle X-ray diffraction (LA-XRD) and nitrogen sorption analyses not only revealed the conservation of the textural properties of the support but also the permeability of the copper Schiff base complex into the channel system despite simultaneous decreases in surface area, pore volume and pore size. Electron paramagnetic resonance (EPR) studies confirmed the presence of copper Schiff base in MCM-41 even after dimerisation with o-aminophenol. The catalytic reaction was carried out with tert-butyl hydroperoxide (TBHP) and aqueous peroxide for MCM-41 and heterogenised copper complex under the mild reaction conditions.


 Please wait while we load your content...
Please wait while we load your content...