The effect of nitrogen and oxygen coordination: toward a stable anode for reversible lithium storage†
Abstract
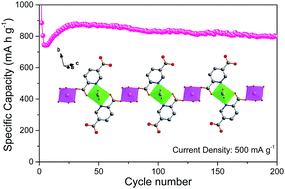
Metal–organic frameworks for lithium storage have gained intensive attention, but their cycling stabilities are still limited. A multi-coordinated strategy to improve the cycling stabilities is explored, inspired by the study of the stability and structure of metal ions coordinated with the amide group. A cobalt-based 2,5-pyridinecarboxylic metal–organic chain, named Co-pydc, was synthesized and applied as an anode for lithium-ion batteries after evacuation. The evacuated Co-pydc shows a unique laminar and porous structure, which benefits the Li-ion diffusion, thus exhibiting a faster activation of the electrode and capacitive-controlled electrochemical behavior. When tested at 100, 500, and 1000 mA g−1 for 50, 200, and 400 cycles, respectively, the evacuated Co-pydc delivered high capacities of 1036.4, 798.6, and 589.2 mA h g−1 after the final cycle, respectively, demonstrating outstanding cycling stabilities. The excellent cycling ability mainly arises from the unique chain network induced by the synergistic multi-coordination of pyridinic nitrogen and carboxylic oxygen.


 Please wait while we load your content...
Please wait while we load your content...