Can molten carbonate be a non-metal catalyst for CO oxidation?
Abstract
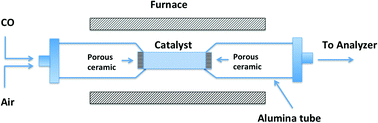
For the first time, we have examined molten carbonate as a non-metal catalyst for CO oxidation in the temperature range of 300–600 °C. The reaction mechanism was analyzed using a classic Langmuir–Hinshelwood model combined with DFT calculations. It was found that the conversion of CO is greatly enhanced by molten carbonate at about 400 °C and increased to 96% at 500 °C. The reaction process involves four steps, including (1) dissociative adsorption of oxygen, (2) adsorption of CO, (3) surface reaction, and (4) desorption of CO2. DFT modeling reveals the formation of (C2O4)2− and (CO4)2− as the intermediate species, and that the first two steps are exothermic and preferred by chemical equilibrium. The energy barrier of oxygen dissociation to form CO42− is calculated to be 23.0 kcal mol−1, which is in good agreement with the measured overall activation energy of 19.1 kcal mol−1. However, the surface reaction (step 3) has a low energy barrier of 10.8 kcal mol−1 only. This confirms that the oxygen dissociation is the rate determing step in the whole process. Further analysis of the reaction kinetics indicates that the reaction is affected by the CO concentration. With low CO concentration, the reaction is first order with respect to CO and half order to O2. From the current report, it has been proven that molten carbonate can serve as an efficient catalyst for CO oxidation and potentially for other oxidation reactions in the temperature range of 400–600 °C. More studies are demanded to further investigate the reaction mechanism and explore more potential industrial applications.


 Please wait while we load your content...
Please wait while we load your content...
