One-pot synthesis of highly fluorescent Fe2+-doped carbon dots for a dual-emissive nanohybrid for the detection of zinc ions and histidine†
Abstract
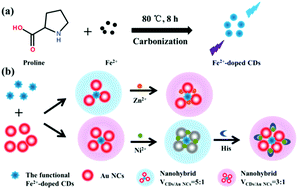
Metal ion-doping has been paid little attention for improving the fluorescence intensity of carbon dots (CDs) compared with nonmetallic-element doping. Herein, for the first time, Fe2+-doped CDs were facilely synthesized at 80 °C instead of by a hydrothermal method. Fe2+ was confirmed to be the only definitive common metal ion to synthesize highly fluorescent CDs with proline (Pro) as a carbon resource. The as-synthesized Fe2+-doped CDs showed bright blue fluorescence at 455 nm and exhibited favorable anti-ion, pH, and time stability. The blue cysteine-functional-Fe2+-doped CDs were combined with red gold nanoclusters (Au NCs) to construct a nanohybrid by electrostatic interaction for the ratiometric fluorescence sensing of Zn2+ and histidine (His), in which the CDs acted as a reference signal and the Au NCs as an analyte-responsive unit. The detection of Zn2+ and histidine (His) was shown by two distinct blue-pink and pink-blue-pink fluorescence color changes, respectively. Both of them showed excellent selectivity and sensitivity. Dual-emission fluorescent probes avoid the shortcomings of single fluorescence signals, which are susceptible to experimental conditions, and have a bright future being utilized for the visual detection of Zn2+ and His in practical analysis.


 Please wait while we load your content...
Please wait while we load your content...