Cycloplatinated(ii) complexes bearing an O,S-heterocyclic ligand: search for anticancer drugs†
Abstract
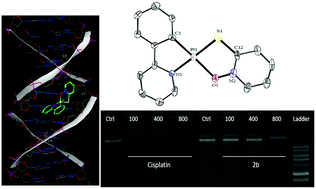
2-Mercaptopyridine N-oxide sodium salt (NaSpyO) was used as a bioactive ligand for the preparation of two new cycloplatinated(II) complexes. The reaction of the NaSpyO ligand with the cyclometalated platinum complexes [PtCl(C^N)(dmso)], 1, C^N = deprotonated form of 2-phenylpyridine (ppy, 1a) or 7,8-benzoquinoline (bzq, 1b), afforded the complexes [Pt(C^N)(SpyO)], 2, C^N = ppy (2a) or bzq (2b). These complexes were characterized in solution via NMR spectroscopy. Single-crystal X-ray determination (in the case of 2a) and theoretical calculations confirmed that the SpyO ligand acted as a chelate (sulfur atom is cis to the carbon atom of the C^N ligand). The optical properties of both complexes were studied by UV-visible spectroscopy. The lower energy bands in 2 had a mixed transition character (1MLCT/1LC/1L′LCT, L = C^N and L′ = SpyO), and this assignment was supported by the time-dependent density functional theory (TD-DFT) calculations. The cytotoxic activities of 2 were tested on five different human cancer cell lines: A549 (non-small cell lung cancer), MCF-7 (breast cancer), SKOV3 (ovarian cancer), MDA-MB-231 (breast cancer), and HT-29 (colon colorectal cancer). The effects of these compounds on the proliferation of the non-tumoral breast cell line (MCF-12A) showed good selectivity among the tumorigenic and non-tumorigenic cell lines. These complexes revealed good to moderate anti-proliferative activity. Electrophoresis mobility shift assays and molecular docking studies were also carried out on 2 to determine the specific binding mode and the orientation of binding to DNA.


 Please wait while we load your content...
Please wait while we load your content...