Chromium-based metallosurfactants: synthesis, physicochemical characterization and probing of their interactions with xanthene dyes†
Abstract
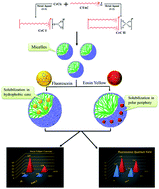
Surfactants are known to enhance the photophysical properties of dyes. In this work, a photophysical study of xanthene dyes in micellar media of single and double alkyl chain cationic chromium-based metallosurfactants has been carried out. For this study, two chromium complexes, i.e. hexadecyltrimethyl ammonium chromium trichlorate and bishexadecyltrimethyl ammonium chromium tetrachlorate, were synthesized and characterized using elemental analysis, Fourier transform infrared spectroscopy, thermogravimetric analysis and nuclear magnetic resonance. Conductivity and surface tension methods were employed to assess the role of the metal ion (which is present as part of the counter-ion) in the self-aggregation behavior of as-synthesized metallosurfactants. Furthermore, the interaction of two chromium metallosurfactants was investigated with xanthene dyes (fluorescein and eosin yellow), wherein binding constants were evaluated using fluorescence and UV-vis spectroscopy. Due to the difference in the structure of the dyes, fluorescein shows a higher binding value than eosin yellow. This may be due to the steric crowding of eosin yellow due to an additional substituent that causes difficulty in the interaction of the apolar group of the dye with the hydrophilic region of the chromium complexes. The solubilization loci of both the dyes were estimated where fluorescein being hydrophobic occupied the hydrophobic core of the metallomicelles and eosin yellow was present in the outer polar Stern region. The double alkyl chain chromium metallosurfactant, being more hydrophobic, exhibits strong binding towards fluorescein and eosin yellow in comparison to the single alkyl chain chromium metallosurfactant. Moreover, chromium metallosurfactants enhance the solubilization and fluorescence quantum yield of the dyes.


 Please wait while we load your content...
Please wait while we load your content...