Mercaptopropionic acid-capped Mn-doped ZnS quantum dots as a probe for selective room-temperature phosphorescence detection of Pb2+ in water
Abstract
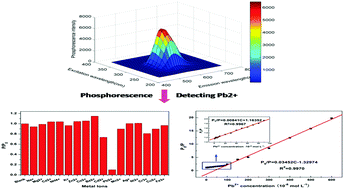
In this study, highly water-soluble Mn-doped ZnS quantum dots (QDs) capped by 3-mercaptopropionic acid (MPA) were successfully synthesized by three different synthetic methods. By comparison, QDs prepared by hydrothermal synthesis method at 100 °C heating for 2 h had the best phosphorescence emission properties. Room-temperature phosphorescence (RTP) could be remarkably and selectively quenched by Pb2+ in a pH 3.8 NaAc–HAc buffer solution. Based on the abovementioned finding, a simple, sensitive, and selective phosphorescence method for rapid detection of Pb2+ was successfully developed using MPA-capped Mn-doped ZnS QDs as a probe. Under the optimized conditions, the RTP intensity ratio of P0/P had a good linear relationship with Pb2+ concentration in the concentration ranges of 4 × 10−8–90 × 10−8 mol L−1 and 90 × 10−8–600 × 10−8 mol L−1 with the correlation coefficients of 0.9967 and 0.9970, which well followed the Stern–Volmer quenching equation, and the Stern–Volmer quenching constants were 8.41 × 105 L mol−1 and 3.452 × 106 L mol−1, respectively. The detection limit was 3.69 × 10−8 mol L−1, and the relative standard deviation for 11 repeated detections of 60 × 10−8 mol L−1 Pb2+ was 2.8%. According to the analysis of the Stern–Volmer quenching equation, UV-Vis absorption spectra, decay curves of phosphorescence emission, and phosphorescence lifetime, the quenching mechanism may imply that energy transfer and charge transfer in the interaction of MPA-capped Mn-doped ZnS QDs at the excited state with Pb2+ made QDs lose excitation energy. This resulted in phosphorescence of QDs causing dynamic quenching. The proposed method was successfully applied to detect Pb2+ in real water samples with satisfactory results, and the recoveries ranged from 93% to 109.3%. The developed method was simple, rapid, and specific and could effectively avoid fluorescence interference of the system, which opened up a promising prospect for the sensitive, convenient, and fast sensing and monitoring of small molecule pollutants in water based on phosphorescence sensors.


 Please wait while we load your content...
Please wait while we load your content...