Enhanced electrochromic switching speed and electrochemical stability of conducting polymer film on an ionic liquid functionalized ITO electrode
Abstract
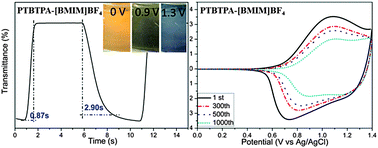
The 1-butyl-3-methylimidazolium tetrafluoroborate ([BMIM]BF4) functionalized ITO substrate was successfully prepared via a solution immersion method and then incorporated with poly(4,4′,4′′-tris[4-(2-bithienyl)phenyl]amine) (PTBTPA) to form the PTBTPA–[BMIM]BF4 film by electrochemical polymerization, which presents reversible multicolor changes from orange, olive green to dark gray. Interestingly, compared with the bleaching time (tb) and the coloring time (tc) of the pure PTBTPA film (1.76 s and 4.51 s) at 1100 nm, the PTBTPA–[BMIM]BF4 film exhibits shorter tb and tc (0.87 s and 2.90 s) at the same wavelength. Obviously, the switching speed of the PTBTPA–[BMIM]BF4 film has been improved significantly, and it is further supported by the electrochemical impedance spectra which demonstrate that the PTBTPA–[BMIM]BF4 film possesses much lower charge transfer resistance. The reduction of charge transfer resistance could be attributed to (1) the private channel provided by the ionic liquid [BMIM]BF4 as a linker between the polymer and the electrode; (2) the ability of the simultaneous doping and dedoping of ClO4− in the electrolyte and BF4− ions of the ionic liquid. Moreover, the cyclic stability studies reveal that the PTBTPA–[BMIM]BF4 film exhibits better durability and retains 70.4% of its original electroactivity after 500 cycles in ionic liquid solution. The results demonstrate that the electrochemical and the electrochromic performances could be significantly enhanced through incorporating PTBTPA with the ionic liquid ([BMIM]BF4).

 Please wait while we load your content...
Please wait while we load your content...