Hg2+-induced deprotonation of an anthracene-based chemosensor: set–reset flip-flop at the molecular level using Hg2+ and I− ions†
Abstract
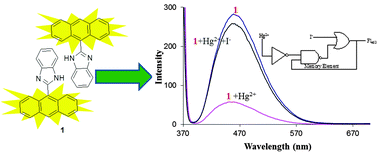
A highly selective, simple, cost-effective anthracene-based chemosensor (1) has been developed. It exhibits an Hg2+-selective “on–off” fluorescence quenching behavior, wherein Hg2+ induces the deprotonation of imidazole NH, as rationalized from 1H NMR titrations. The system exhibits a fluorescence color change from fluorescent bluish-green to blue with high selectivity and sensitivity over other metal ions. The in situ generated deprotonated species of 1 with Hg2+ can recognize I− ions via fluorescence enhancement. The present sensing system is successfully applied for the detection of Hg2+ ions in real samples. Chemosensor 1 can also mimic the functioning of set–reset flip-flop with chemical inputs from Hg2+ and I− ions.

 Please wait while we load your content...
Please wait while we load your content...