Synthesis and optical properties of phenanthromidazole derivatives for organic electroluminescent devices†
Abstract
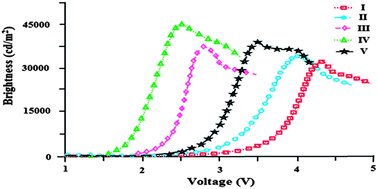
Five phenanthromidazole derivatives, namely 2-(naphthalen-1-yl)-1H-phenanthro[9,10-d]imidazole (1), 2-(naphthalen-1-yl)-1-phenyl-1H-phenanthro[9,10-d]imidazole (2), 2-(naphthalen-1-yl)-1-p-tolyl-1H-phenanthro[9,10-d]imidazole (3), 1-(4-methoxyphenyl)-2-(naphthalen-1-yl)-1H-phenanthro[9,10-d]imidazole (4) and 1-(3,5-dimethylphenyl)-2-(naphthalen-1-yl)-1H-phenanthro[9,10-d]imidazole (5), were synthesised using the TiO2 (R) nanosemiconductor as the catalyst under solvent free conditions and characterised by NMR and single crystal XRD techniques. Their photophysical, electrochemical and electroluminescent properties were carefully analysed. Highly efficient Alq3-based organic light emitting devices have been developed using phenanthromidazoles as functional layers between NPB [4,4-bis(N-(1-naphthyl)-N-phenylamino)biphenyl] and Alq3 [tris(8-hydroxyquinoline)aluminium] layers. Using the device of ITO/NPB/4/Alq3/LiF/Al, a maximum luminous efficiency of 5.99 cd A−1 was obtained with a maximum brightness of 40 623 cd m−2 and a power efficiency of 5.25 lm W−1.

 Please wait while we load your content...
Please wait while we load your content...