Synthesis, X-ray structural features, DFT calculations and fluorescence studies of a new pyridoxal-benzimidazole ligand and its respective molybdenum complex†
Abstract
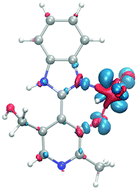
Pyridoxal hydrochloride reacts with 1,2-phenylenediamine to give 4-(1H-benzimidazol-2-yl)-5-(hydroxymethyl)-2-methylpyridin-3-ol hydrochloride (BIMIPY, 1). The reaction of 1 with bis(acetylacetonato)-dioxidomolybdenum(VI) leads to the formation of the complex [(MoO2)2(H2O)2(μ-O)(BIMIPY–H+)2]·4DMSO·2H2O (2). In the binuclear complex the metal centers attain a distorted octahedral geometry; a water molecule coordinates to each Mo atom and two imidazolic ligands chelate two MoO2+2 ions. The sixth coordination position in complex 2 is accomplished by a μ-oxido ligand, with an inversion center, which connects two moieties of [(MoO2)(H2O)(BIMIPY–H+)] to a dinuclear species. Secondary, intermolecular hydrogen bonds support the growth of the supramolecular assembly of the Mo complex. Mechanisms for the formation of the functionalized benzimidazole ligand are also proposed and discussed. First principles DFT calculations of reactivity indexes are used to investigate a possible mechanism leading from the benzimidazole to a dimeric complex involving (MoO2+2) molecules and an oxido bridge ligand. Further, the expected fluorescence of the complex is absent, due to decomposition of the complex in solution. On the other hand, an experimental investigation of the photophysical properties of the ligand shows that it is fluorescent. Time dependent density functional theory calculations are performed to characterize the absorption and fluorescence spectra of the ligand.

 Please wait while we load your content...
Please wait while we load your content...